Nuclear Medicine
Nuclear medicine images function, not just structure. By administering radioactive tracers that accumulate in specific tissues, it reveals metabolic activity, receptor density, and physiologic processes invisible to other modalities.
Radioactive Decay & Imaging
Unstable atomic nuclei spontaneously emit radiation as they decay toward a more stable state. Nuclear medicine harnesses this phenomenon by introducing radiolabeled compounds (radiopharmaceuticals) into the body that are taken up by specific tissues.
The key types of decay relevant to nuclear medicine imaging are gamma (γ) decay (for SPECT and planar scintigraphy) and positron (β⁺) emission (for PET scanning).
- Gamma decay: Nucleus emits a high-energy photon (gamma ray) without changing atomic number. Gamma rays pass through tissue and are detected externally by the gamma camera.
- Positron emission: A proton converts to a neutron, emitting a positron (β⁺). The positron travels a short distance, then annihilates with an electron to produce two 511 keV photons traveling in exactly opposite directions — detected by PET coincidence detection.
- Half-life (T½): Time for half the radioactive atoms to decay. Short T½ = less patient dose but requires fast imaging; long T½ = sustained imaging but more dose.
- Effective half-life: Combined physical decay AND biological elimination — true dose predictor in vivo.
Radiopharmaceuticals
A radiopharmaceutical = radioactive isotope + pharmaceutical carrier. The pharmaceutical determines where the tracer accumulates in the body; the isotope provides the detectable radiation.
| Radioisotope | Half-Life | Emission | Energy | Key Use |
|---|---|---|---|---|
| Tc-99m | 6 hours | Gamma (γ) | 140 keV | Bone scan, thyroid, cardiac, renal, lung (V/Q) |
| I-123 | 13 hours | Gamma | 159 keV | Thyroid imaging, SPECT brain (DATscan) |
| I-131 | 8 days | Beta (β⁻) + Gamma | 364 keV γ | Thyroid cancer therapy, remnant ablation |
| Ga-67 | 3.3 days | Gamma | 93–394 keV | Infection/inflammation, lymphoma |
| Tl-201 | 73 hours | X-ray + Gamma | 69–83 keV | Cardiac perfusion (older), parathyroid |
| F-18 FDG | 110 min | Positron (β⁺) | 511 keV pairs | PET oncology, neurology, cardiology |
| Ga-68 | 68 min | Positron (β⁺) | 511 keV pairs | PET PSMA (prostate), somatostatin receptors (NETs) |
| Lu-177 | 6.7 days | Beta (β⁻) + Gamma | 208 keV γ | PRRT therapy (NETs), PSMA therapy (prostate) |
Half-Life Activity Calculator
Change the isotope, starting activity, and elapsed time to see how much radioactivity remains. Add biological clearance to estimate effective half-life in the body.
Animated Radioactive Source
The source emits fewer photons as fewer unstable nuclei remain. Lower activity means weaker glow, fewer detected events, and less dose rate over time.
Why Tc-99m dominates: Its 6-hour half-life is long enough for imaging but short enough to minimize patient dose. Its 140 keV gamma energy is ideal for gamma camera detection. It is produced from Mo-99/Tc-99m generators in hospital radiopharmacies, making it readily available. It can be tagged to dozens of different pharmaceutical carriers for different organ systems.
Click components to learn their function.
Gamma Camera Components
Invented by Hal Anger in 1958, the Anger scintillation camera remains the core detector in nuclear medicine. Click components to learn each layer's function.
The gamma camera detects gamma photons emitted by radiopharmaceuticals inside the patient, creating a 2D projection image (planar scintigraphy) or 3D SPECT images through rotation.
SPECT vs PET
Both SPECT and PET create 3D images of radiopharmaceutical distribution. They differ in the type of isotope used, detection method, and image quality.
Visual Comparison
Side-by-side detector geometry, photon emission, tracer examples, and typical image results for SPECT and PET.
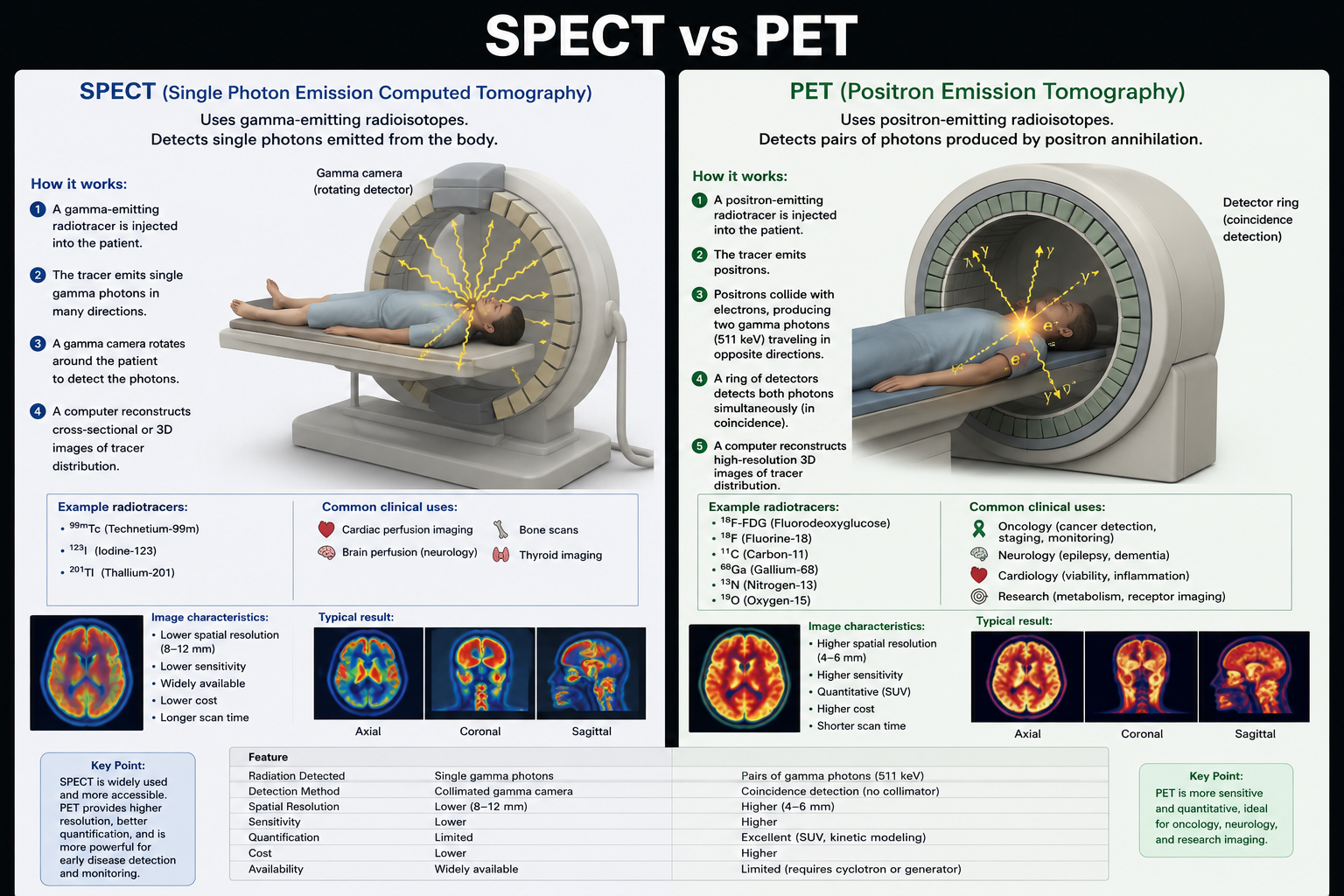
| Feature | SPECT | PET |
|---|---|---|
| Full name | Single Photon Emission CT | Positron Emission Tomography |
| Isotope type | Gamma emitters (Tc-99m, I-123) | Positron emitters (F-18, Ga-68, C-11) |
| Detection method | Rotating gamma cameras + collimators | Coincidence detection (two 511 keV photons) |
| Spatial resolution | 8–12 mm | 3–5 mm (superior) |
| Sensitivity | Lower | 10–100× higher than SPECT |
| Quantification | Semi-quantitative (SUV limited) | Accurate quantification (SUV) |
| Scan time | 20–60 min (rotating) | 10–30 min (stationary ring) |
| Cost | Lower (Tc-99m from generator) | Higher (cyclotron needed for most tracers) |
| Hybrid scanner | SPECT/CT | PET/CT or PET/MRI |
| Main clinical use | Bone, cardiac, thyroid, renal, lung | Oncology (FDG-PET), neurology (amyloid), cardiology |
PET/CT fusion: Combining a PET scanner with a CT scanner in one unit (PET/CT) allows simultaneous functional (PET) and anatomical (CT) imaging in a single session. The CT provides anatomical localization and attenuation correction for the PET data — this combination revolutionized oncology staging and treatment response assessment.
Clinical Applications
Nuclear medicine uniquely reveals functional information — metabolism, perfusion, receptor density — that anatomical imaging cannot provide.
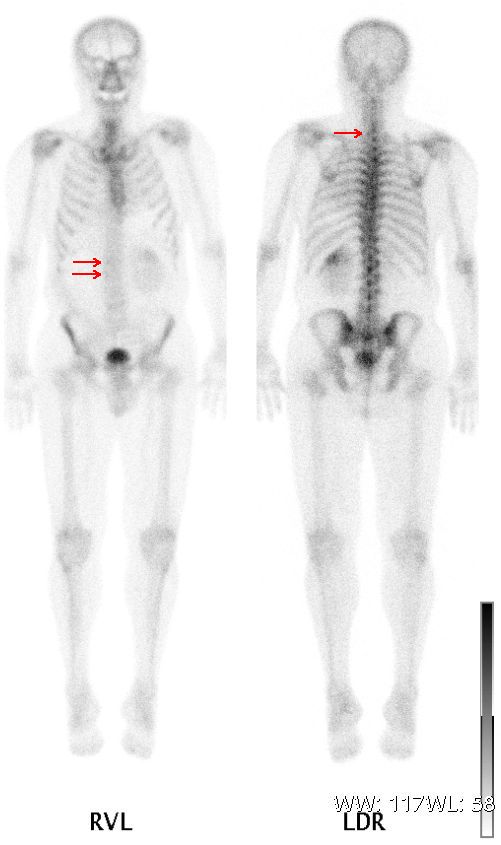
🦴 Bone Scan (MDP Tc-99m)
Whole-body bone scan for metastases, stress fractures, osteomyelitis, and Paget's disease. Highly sensitive for bone turnover — detects lesions earlier than plain X-ray.

❤️ Myocardial Perfusion (MPI)
SPECT with Tc-99m sestamibi or Tl-201 to assess coronary artery disease. Stress/rest comparison identifies ischemia vs. infarct. PET with Rb-82 or NH3 for quantitative perfusion.
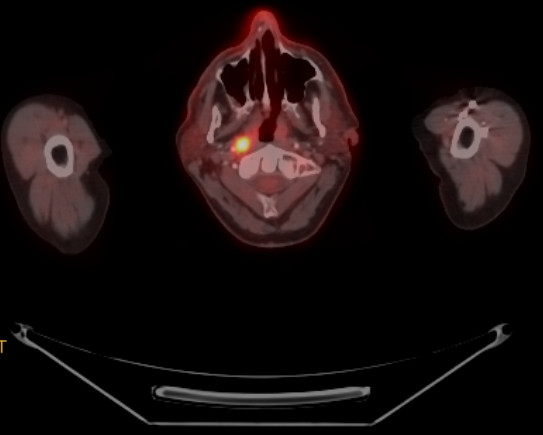
🎗️ FDG-PET Oncology
F-18 fluorodeoxyglucose (FDG) exploits the Warburg effect (increased glucose metabolism in cancer). Used for staging, restaging, and treatment response in lymphoma, lung, colorectal, melanoma, and many other cancers.
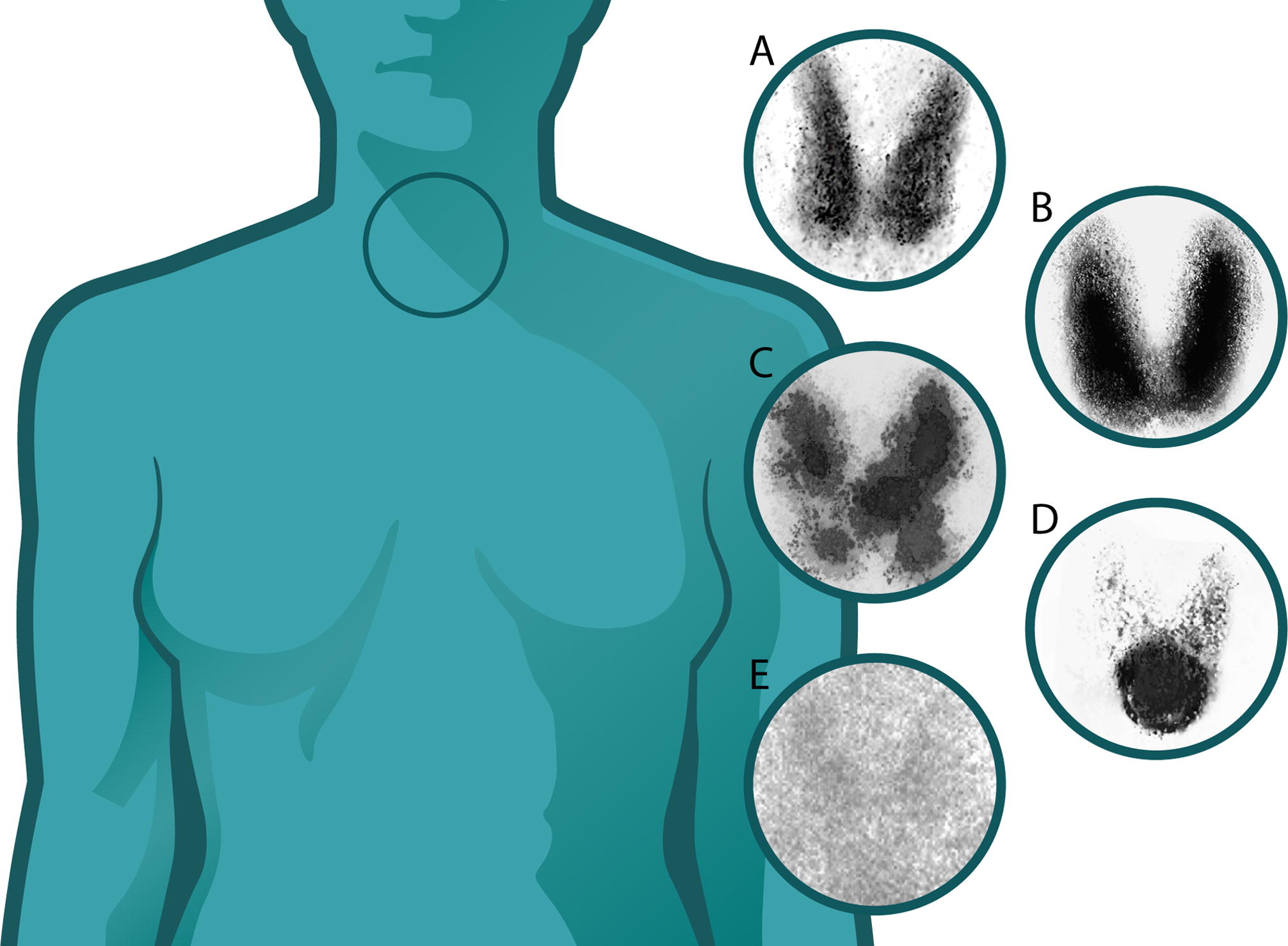
🦋 Thyroid Studies
I-123 thyroid scan: hot (autonomous) vs. cold (suspicious) nodules, Graves' disease uptake. I-131 treatment and ablation for thyroid cancer and hyperthyroidism.
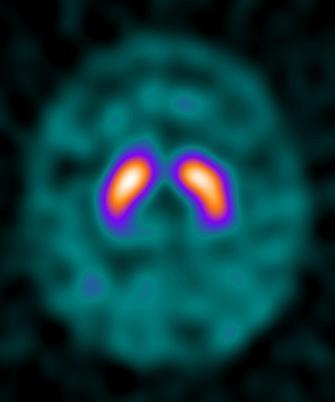
🧠 Neurology PET
FDG-PET for dementia (Alzheimer's vs FTD pattern). Amyloid PET (florbetapir) and tau PET for Alzheimer pathology. DATscan (I-123) for Parkinson's vs. essential tremor.
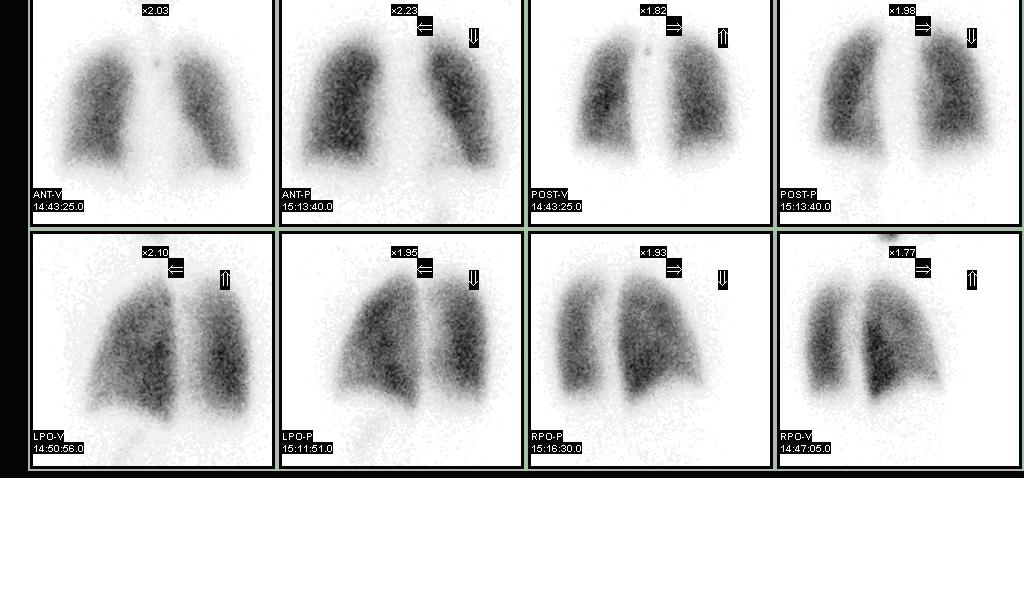
🫁 V/Q Scan (Lung)
Tc-99m MAA perfusion + Tc-99m aerosol or Xe-133 ventilation for pulmonary embolism diagnosis. Preferred over CT in pregnancy (lower fetal dose).
Radiation protection for NM staff: Time, distance, and shielding are key. Keep exposure times minimal during radiopharmaceutical preparation. Tungsten syringe shields and lead-lined waste containers are standard. Pregnant patients — assess risk/benefit; most studies avoided in first trimester.
Radiation as Treatment
Radiotherapy — harnessing high-energy radiation to destroy cancer cells.
Explore Radiotherapy →